NCCIH Training and Career Development Opportunities
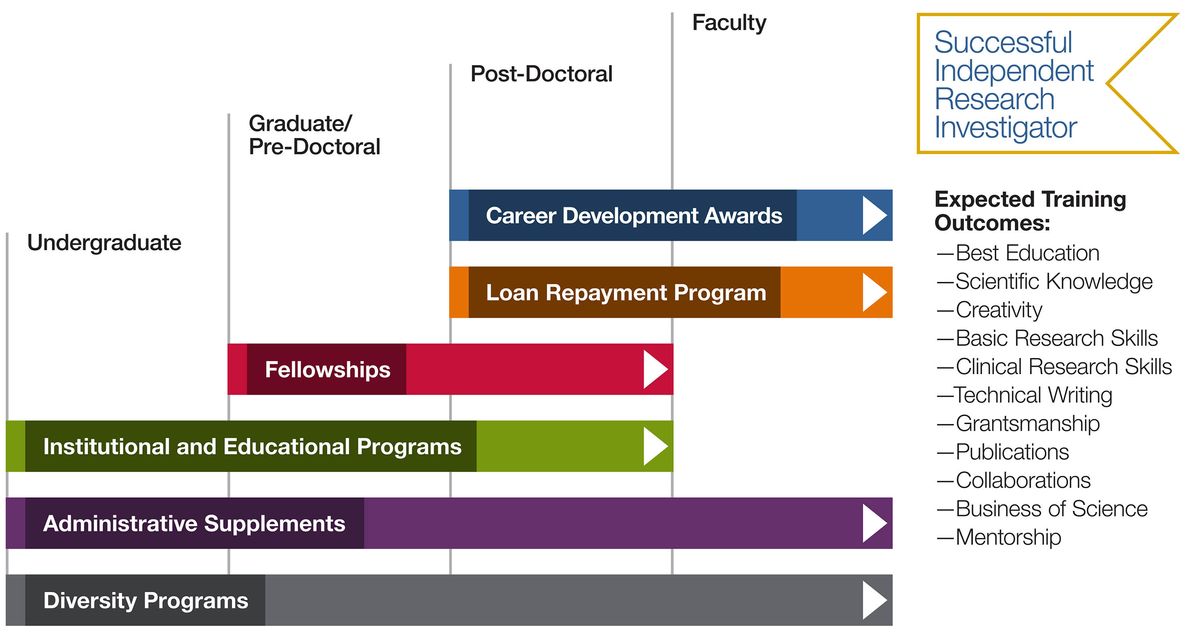
Click on the program names below to see more information.
For More Information
- About Research Training and Career Development
- NCCIH-Funded Institutional Research-Training Programs
- Grant Application, Review, and Award Processes
- More Training Resources
- Research FAQ (NIH)
Contacts
For questions and/or to discuss the scientific area of your research project:
Please contact Jennifer Baumgartner, Ph.D., for questions regarding fellowships or the Loan Repayment Programs.
Please contact Lanay Mudd, Ph.D., for all other programs.
For questions about grants management and budget:
Debbie Chen
Chief Grants Management Officer
Email: debbie.chen@nih.gov